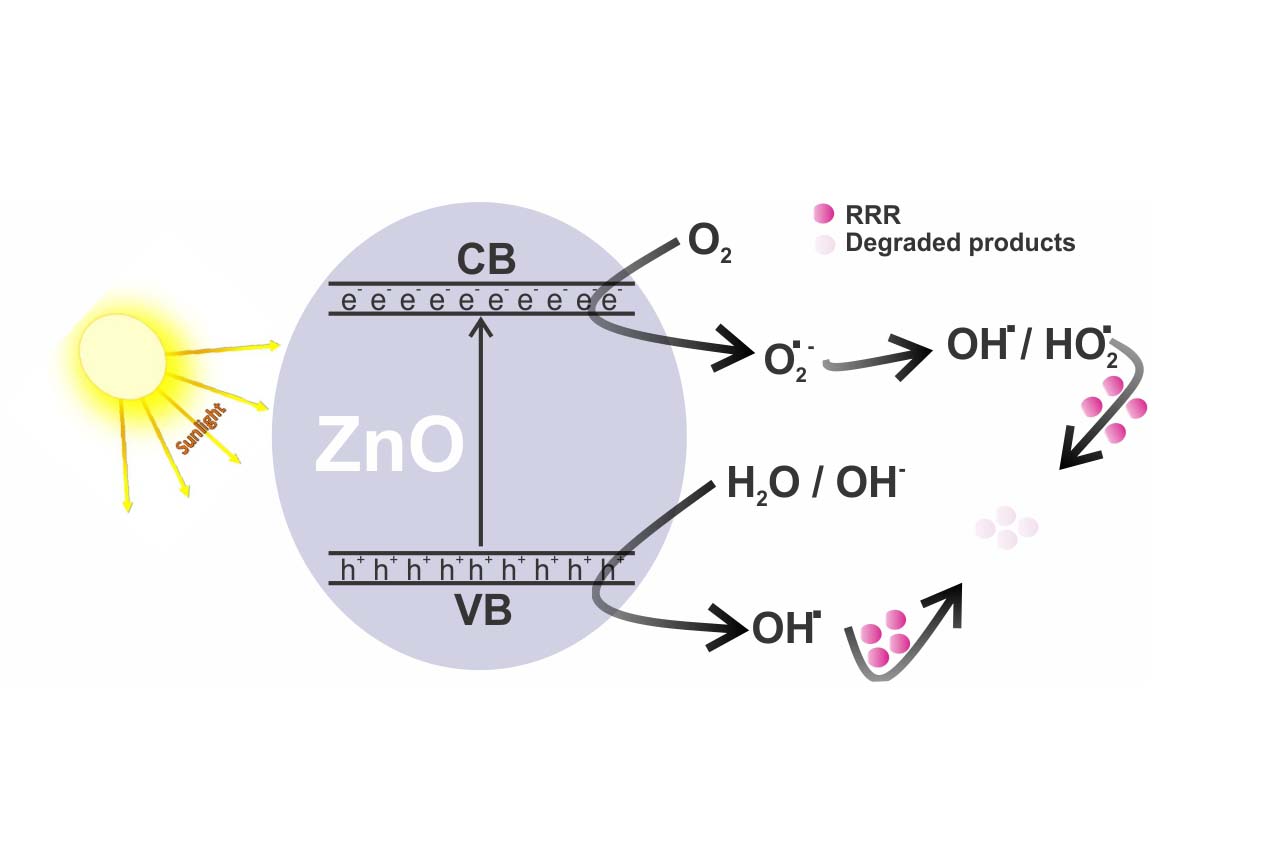
ABSTRACT: The nanoparticles of ZnO (n-ZnO) have been synthesized by a sol-gel method and characterized by UV-visible and FTIR spectroscopy, scanning electron microscopy (SEM), energy dispersive X-Ray spectrometry (EDX) and powder X-ray diffractometry (XRD). Precursor of n-ZnO particles were prepared via a non-aqueous route, which was calcined at 500oC. These particles were then deposited on a glass substrate for adsorption and photodegradation of a typical textile dye, Remazol Red RR (RRR). Especially, the high surface to volume ratio of nanoparticles has appealed much attention to use these particles both as an adsorbent and a photocatalyst. A comparative study was carried out between n-ZnO and a commercially available ZnO (c-ZnO) to investigate the removal efficiency of RRR from its aqueous solution under different conditions. The removal efficiency has been optimized by varying several operating variables and the highest performance has been obtained with 0.115 g/slide of ZnO and 0.5 × 10-4 M aqueous solution of RRR under sunlight irradiation. It is important to note that the use of the films of ZnO in the presence of solar light makes it suitable for recycling and causes no secondary environmental pollution.
Keywords: nanoparticles, ZnO film, heterogeneous photocatalysis, Remazol Red RR, adsorption, photodegradation
